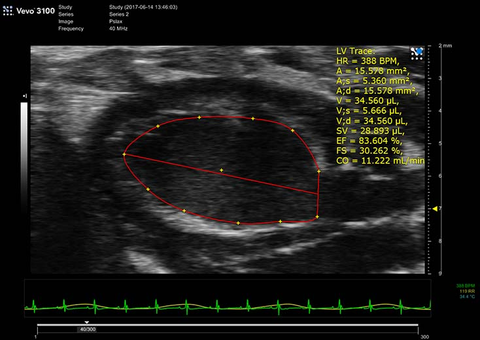
PSLAX LV Trace B-Mode Diastole

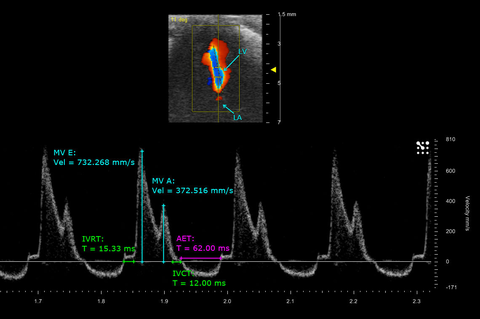
Mitral flow and measurements of a mouse neonate heart

PW Doppler of mitral blood flow in a postnatal day 9 mouse taken from an apical 4 chamber view, with related cardio measurements.

Parasternal short axis view of mouse neonate heart
Left ventricle of neonatal mouse at postnatal day 4, imaged in parasternal short axis view, with papillary muscles labeled.

Mouse neonate heart PND 4
Parasternal Long Axis view of a mouse neonate heart imaged at postnatal day 4. LV, left ventricle. RVOT, right ventricular outflow track. Ao root, aortic root.

Parametric Map of Oxygen Saturation in an Orthotopic Breast Tumor Model
In this parametric map of oxygen saturation in an orthotopic breast tumor, the breathed oxygen concentration is varied from 100% to 5% back to 100% and the resulting changes in sO2 can be observed in different areas.
Note that the sO2 in the tumor interior does not change appreciably during the breathed oxygen challenge, indicating impaired function of the vasculature there.
Oxygen saturation in tumor

Perfusion in Tumor

3D rendered high-resolution ultrasound (greyscale) and nonlinear contrast (beige) image of a subcutaneous tumor showing perfusion in the tumor tissue.
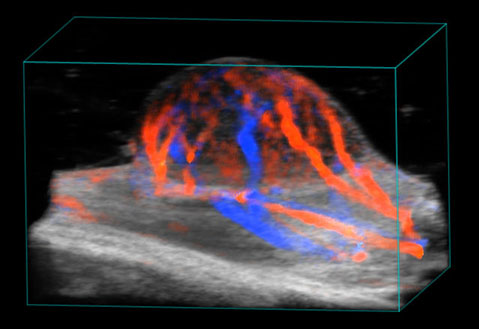
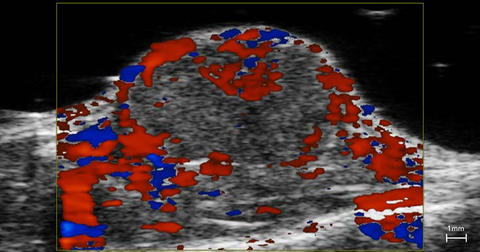
Blood flow in tumor

3D rendered high-resolution ultrasound (greyscale) and color Doppler (orange and blue) image of a subcutaneous tumor showing blood flow.
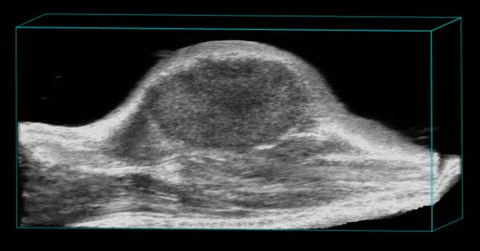
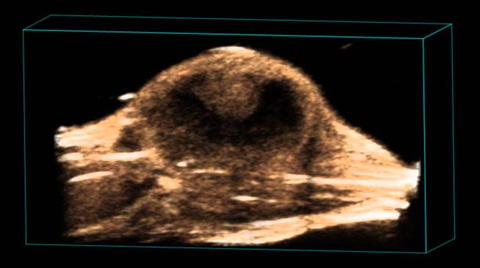
Subcutaneous tumor

3D rendered high-resolution ultrasound image of a subcutaneous tumor.
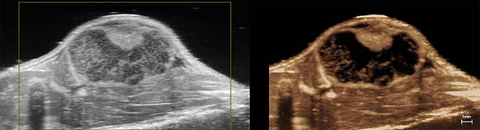
Nanoparticle distribution in tumor

High-resolution ultrasound (left) and spectrally unmixed photoacoustic (right) image of a subcutaneous tumor showing nanoparticle distribution (yellow) as well as oxygenated (red) and deoxygenated (blue) hemoglobin signal.
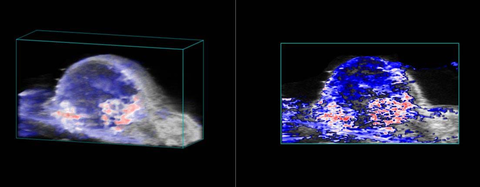
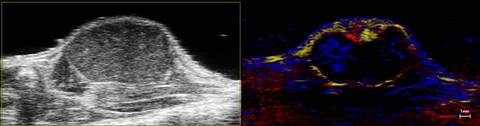
Perfusion in Tumor

High-resolution ultrasound (left) and nonlinear contrast (right) image of a subcutaneous tumor showing perfusion in the tumor tissue.
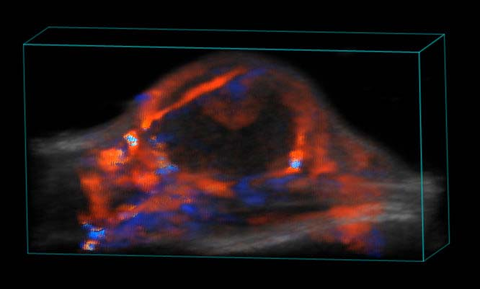
Blood flow in tumor

Perfusion in Tumor

3D rendered high-resolution nonlinear contrast image of a subcutaneous tumor showing perfusion in the tumor tissue.
Blood flow in tumor

3D rendered high-resolution ultrasound (greyscale) and color Doppler (orange and blue) image of a subcutaneous tumor showing blood flow.
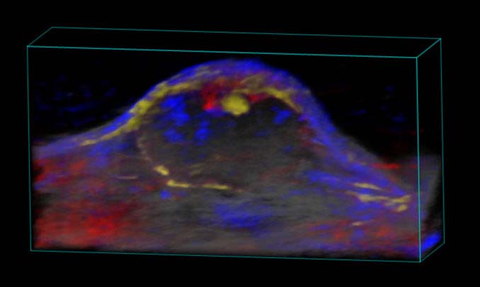
Nanoparticle distribution in tumor

3D rendered high-resolution ultrasound (greyscale) and spectrally unmixed photoacoustic (red, blue and gold) image of a subcutaneous tumor showing nanoparticle distribution (yellow) as well as oxygenated (red) and deoxygenated (blue) hemoglobin signal.
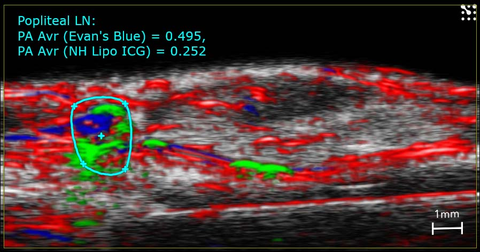
Imaging of lymphatics in the mouse.

High-resolution ultrasound (greyscale) and photoacoustic (red, blue and green) image of the mouse hindlimb showing Evan's Blue (blue) and ICG (green) dye in the lymphatic vessels and the popliteal lymph node.
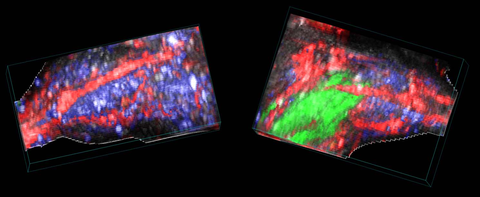
Stem cells in the mouse hindlimb

3D rendered spectrally unmixed photoacoustic image of a stem-cell injected, ischemic (right) and control (left) mouse hindlimb. Oxyhemoglobin (red), deoxyhemoglobin (blue) and dye-labelled stem cells (green) are shown.
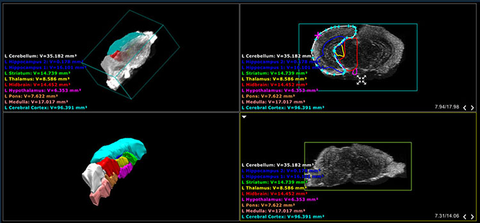
Screenshot of mouse brain atlas

Screenshot of the Vevo LAB software displaying several different views of the CD-1 mouse brain atlas which comes with the Vevo BRAIN stereotactic frame.
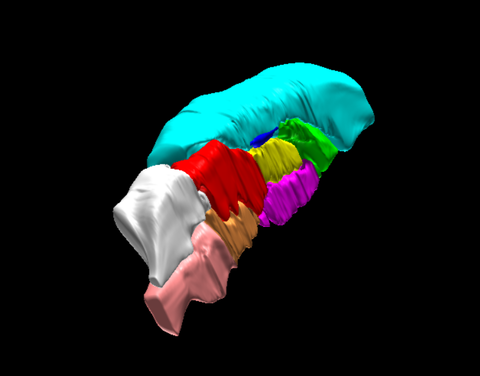
Neuroanatomy of the mouse brain

3D rendered surface view of the mouse brain segmented by anatomical region based on ex vivo high frequency ultrasound images.
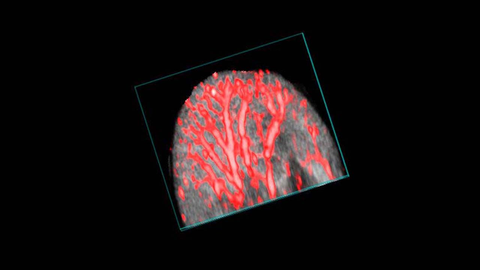
Mouse ear vasculature

3D rendered coregistered ultrasound (greyscale) and photoacoustic (red) image of the mouse ear. The photoacoustic signal is derived from the absorption of light by hemoglobin.
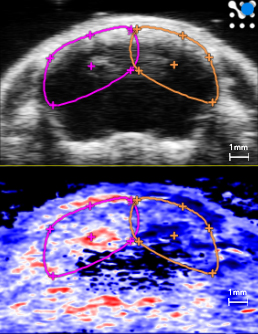
Ischemia/hypoxia stroke model

Plot of oxygen saturation (sO2) over time in the left (pink) and right (orange) cortical and subcortical hemisphere ROIs while restoring breathed oxygen to 100% from 7.5% post ischemia/hypoxia for stroke induction. Ultrasound (top) and photoacoustic (bottom) coronal images in the right panel show a frame of mouse brain post ischemia/hypoxia. The red, white and blue color represents a parametric map of oxygen saturation. The right hemisphere does not fully recover sO2 levels post-reperfusion/normoxia that are shown in the left hemisphere, indicating a cerebral infarct, especially in cortical and subcortical areas.